| Metallic alloys are frequently used under high thermal, chemical and mechanical stress. Numerous energy productions applications favor their oxidation, leading to a degradation of their properties. An extreme case is the nuclear power production, where zirconium based alloys are employed in nuclear reactors, at high temperature, under high humidity, and irradiation levels. The aeronautics industry is also concerned by the corrosion phenomena of alloys used in aircraft gas turbines that decrease their service life and/or their efficiency.
Thermogravimetric analysis (TGA) is a well-established experimental method to study the growth of oxide layers on metallic alloys and can be employed to study corrosion processes providing that the thermobalance used:
This is the case of several TGA designed and manufactured by SETARAM Instrument, more specifically SETSYS Evolution TGA and the symmetrical high temperature thermobalance TAG. |
||||
METHODS
|
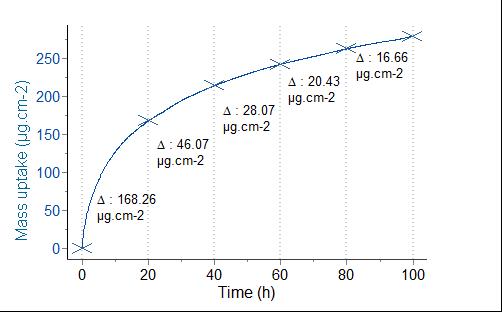 Figure 1 – mass uptake vs. time of an alloy plate tested at 1100°C under a flow of oxygen (30 mL/min) during 100 hours with a SETSYS Evolution TGA |
Cyclic oxidation
When testing the resistance to oxidation at high-temperature, the cyclic-oxidation may be preferred to replicate conditions close to the actual conditions of use. Indeed, it integrates isothermal oxidation kinetics, oxide-scale adherence, mechanical stresses, oxide creep and the evolution of these properties with time.
Complex temperature profiles such as the one depicted by the red curve on Figure 2 can be easily programmed. The result is a stepwise mass increase due to the successive oxidation rate increases during heating and decreases during cooling. A chart showing the mass variation per step as a function of the cycle number can then be derived from the mass uptake signal.
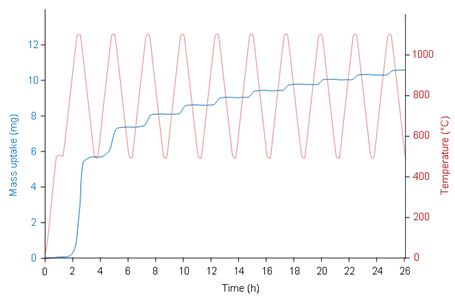 Figure 2 – mass uptake vs. time of a steel plate cycled between 500 and 1100°C under a flow of air (30 mL/min) with a TAG symmetrical TGA. |
Wet atmosphere
In most cases, humidity promotes corrosion, and experiments under dry conditions may not describe accurately the oxidation processes as they occur in wet atmospheres. This is why it is important to mention that SETARAM thermogravimetric analyzers can be connected to the WETSYS humid gas generator.
WETSYS is designed for any application in which controlling the relative humidity rate of a chamber is necessary. It is a compact and automated wet gas generator based on a simple principle: the mixing of a dry gas and a water-saturated gas so as to maintain the relative humidity of the generated outlet gas at a given temperature.
Both isothermal and cycling temperature profiles can be operated under wet conditions as the TGA temperature control is independent of the WETSYS.
 Figure 3 – Wetsys humid gas generator Figure 3 – Wetsys humid gas generator |
APPLICATIONS
Isothermal, dry conditions
Nb-silicide composites are potentially interesting materials for applications in gas-turbine engines for aircraft, where they may face high-temperature (1150°C) hostile environments (oxygen, water vapor, etc). The team of Michel Vilasi from the Institut Jean Lamour in Nancy, France used their TAG symmetrical thermobalance in order to conduct isothermal oxidation tests at 1100°C under an industrial air flow of 1.5 L.h-1 [1]. The parabolic rate constant that is used in oxidation models could be deduced from these tests.
Temperature cycling, dry conditions
The team of Daniel Monceau, from Institut Carnot CIRIMAT in Toulouse, France, has developed a methodology based on temperature cycles meant to determine the Net Mass Gain, the Gross Mass Gain, the total mass of spalled oxide, the rate of metal consumption and the average oxide scale thickness as a function of the number of cycles.
The instruments involved were a standard TAG symmetrical thermobalance and a home designed 6 samples thermobalance system with a six halogen-lamps-furnace and 6 independent alumina sample holders and 6 balances.
They validated their method on slow and fast oxidation kinetics alloy samples, respectively single-crystal beta-NiAl (168 cycles between 150 and 1150°C) at and P91 steel (13 cycles between at 140 and 800°C) [2].
Later on, a custom designed 5 samples thermobalance developed by SETARAM was adapted to a lamps furnace by CIRIMAT and the same method was also applied other materials like Thermal Barrier Coating [3].
Isothermal, wet conditions
Chromium-bearing steels are used as piping or heat exchanger materials in oxyfuel processes for coal combustion, where they are exposed to hot CO2–H2O gas mixtures. The team of Daniel Monceau, from Institut Carnot CIRIMAT in Toulouse, France, has worked on model Fe–Cr and Fe–Cr–Ni alloys, reacted in Ar–CO2 and Ar–CO2–H2O gas mixtures at 650 °C in a Setsys Evolution TGA equipped with a WETSYS [4]. Oxidation isotherms as long as 300 hours, showing mass uptakes as low as 3mg.cm-2 were collected and showed that during reaction in Ar–CO2 and Ar–CO2–H2O at 650 °C, breakaway occurred via nucleation and growth of Fe-rich oxide nodules.
 Figure 5 – Setsys Evolution TGA Figure 5 – Setsys Evolution TGA |
Innovating approaches
At the Saint Etienne School of Mines, in the SPIN lab, an innovative setup involving the coupling between a TAG symmetrical thermogravimetric analyzer and acoustic emission was used to study the Zircaloy-4 corrosion mechanism [5].
The system, in which piezoelectric sensors were placed in the balance head and specific wave guides were employed to hold the samples in the TGA furnace, allowed simultaneous measurements of the sample mass variation and AE burst. Combined with post mortem characterization of oxidized samples, it provided additional information on the Zircaloy-4 corrosion mechanism:
- Kinetic transition is detected under air oxidation tests at 900°C by a change in the rate of mass gain and by the AE activity
- AE allowed distinguishing the cracks which occur during oxidation from the cracks linked with the cooling of the samples


